
“For the first time in the history of food allergy, a large-scale, late-stage clinical trial has shown that oral immunotherapy, done with our investigational medicine, AR101, significantly increased the amount of peanut protein that a patient could consume with no more than mild symptoms,” CEO Stephen Dilly said on a call to investors on Feb. 20.
At the end of the one-year study, about two-thirds of peanut-allergic patients were able to eat 600 milligrams, or approximately two peanuts, which was considerably more than the placebo group. An impressive number were able to tolerate even more at one sitting.
The company plans to file for approval with the FDA at the end of 2018 and, if approved, begin rolling out the treatment midway through 2019.
With AR101, patients eat a carefully processed and measured amount of peanut powder (held in a capsule, then opened and sprinkled in food) on a daily basis. The amount of protein powder in the capsule starts out minuscule, and gradually increases until it reaches a maintenance dose amount of 300 mg, or approximately a peanut.
In the Phase 3 trial, dubbed PALISADE, patients worked up to the maintenance dose of 300 mg for about six months, and then stayed on that dose for another six months. At that point they were evaluated with an oral food challenge by a clinician who had not been involved in their treatment thus far, to minimize any chance of bias.
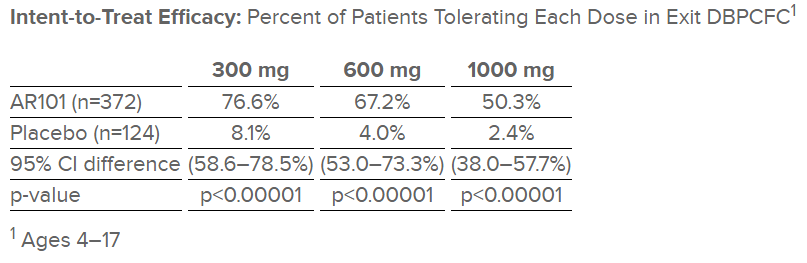
Out of the 496 patients treated in the study, 372 were in the active treatment arm, while the other 124 were given a placebo. Of the 372 patients on AR101, 67 percent tolerated a single dose of 600 mg at the end of the study, or about two peanuts. Only 4 percent of the placebo arm tolerated 600 mg.
 AR101 capsules by dose size. From left: 0.5 milligrams, 1 mg, 10 mg, 20 mg and 100 mg. For maintenance, there are 300 mg sachets.
AR101 capsules by dose size. From left: 0.5 milligrams, 1 mg, 10 mg, 20 mg and 100 mg. For maintenance, there are 300 mg sachets. The patients chosen for the study were considered highly sensitive – almost three-quarters had a history of anaphylaxis, more than half had asthma, and two-thirds had multiple food allergies. None of the patients were able to tolerate more than 30 mg, or one-tenth of a peanut, at the beginning of the study.
Over the course of the treatment, about 20 percent of patients on AR101 dropped out, a rate that’s consistent with other oral immunotherapy trials. Twelve percent left the study due to adverse events, with the most common reason being gastrointestinal symptoms. Ten patients (2.7 percent) experienced systemic reactions, including one case of severe anaphylaxis requiring epinephrine and an overnight hospital stay. The other 8 percent left for non-study-related reasons, including moving out of the area, study fatigue or school commitments.
On the patients who experienced reactions, Dilly emphasized: “This is a drug to be used by allergists who know who to manage the spectrum of allergic reactions as part of their routine practice of desensitization therapy.”
When you look only at the patients who completed the treatment, the therapy looks more successful. A full 96 percent were able to tolerate the equivalent of one peanut, 85 percent were able to tolerate the equivalent of two peanuts, and 63 percent were able to tolerate the equivalent of three peanuts. The company believes that the number of patients who will tolerate higher amounts of peanut will go up the longer they are on the maintenance dose.
Data from two other ongoing Phase 3 trials, one an extension of PALISADE and the other looking at AR101 in real-world clinical settings, is required for AR101’s submission to the FDA. The company is also looking at a much younger study population, as well as adults, where they are so far also seeing favorable results.
 Credit: Aimmune Therapeutics
Credit: Aimmune Therapeutics 2019: Panel Recommends FDA Approve Aimmune’s Peanut Therapy
Hope Remains for the Viaskin Peanut Patch
Aimmune Therapeutics’ February 20, 2018 press release
Backgrounder: OIT Gets Ready for Prime Time





