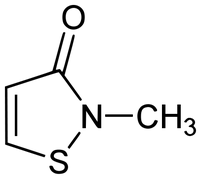
Methylisothiazolinone (or MI) is a preservative used to extend shelf life in a wide variety of cosmetic products. Earlier this year, dermatologists in Britain announced that this chemical was causing allergies at an alarming rate, with positive patch tests to the chemical soaring to 10 percent. (The typical rate of reaction to cosmetics in U.K. patch tests is closer to 1 or 2 percent.)
Even worse is the fact that the prevalence of MI allergy could still be on the rise: “It is a new phenomenon, and at this present time, there is no suggestion that we have reached the top of that frequency or that it is starting to drop,” Dr. Ian White, a leading London dermatologist, told the BBC. “Contact allergy to this permitted preservative is now of epidemic proportions.”
The Johnson & Johnson reformulation was spurred by over 150 complaints to the BBC’s Watchdog program, which is an avenue for consumers to report issues with products and services.
The company, which previously had said the preservative was safe as it was only present in amounts which followed European Union guidelines, has now agreed to reformulate their “Piz Buin 1 Day Long” sunscreen so that it no longer contains MI. They will also gradually remove MI from their current products, and any new products will not contain the allergenic preservative.
“We are continuously seeking to improve the effectiveness and consumer experience of our products,” Johnson & Johnson said in a statement. “As part of this ongoing process we are currently updating Piz Buin 1 Day Long and, from summer 2014, a new formulation that does not contain MI will be available in the UK.”
It is not clear whether Johnson & Johnson plans to remove MI from its North American products. A Canadian spokesperson noted “We are currently evaluating our use of MI and going forward, we have limited its use in new products. MI is currently not being used in new products that are left on the skin and we are evaluating its use in new products that rinse off, such as shampoos and washes.” Requests to the American branch of the company weren’t immediately answered.
U.K. cosmetics retailer Molton Brown has said it was also beginning to remove MI from its products as of September, 2013. Other companies have claimed the chemical is perfectly safe, and will likely wait until legislation is passed requiring them to remove or reduce the ingredient.
See also:
Skin Allergy Epidemic Linked to MI Chemical





