
Updated April 5, 2022: In the U.S., 65.7 percent of adults and kids have received two doses of COVID-19 vaccine and are considered fully vaccinated. As the country contends with newer COVID-19 variants, many people have also received booster shots.
The majority of more severe coronavirus cases continue to emerge among those who aren’t vaccinated or fully vaccinated. While there are several reasons for vaccine hesitation, some people express concern about either vaccine side effects, or the potential for severe allergic reactions.
Allergists say it cannot be repeated often enough – all three vaccines available in the United States – carry a very low risk of anaphylaxis, the severe form of allergic reaction. The CDC advises that the mRNA vaccines and Johnson & Johnson vaccine are safe for those with food allergies and most drug allergies.
A national study published by the U.S. Centers for Disease Control and Prevention (CDC) in 2021 concluded that anaphylaxis occurs in only 2 to 5 people per one million vaccinations given with the Pfizer-BioNTech or Moderna mRNA vaccines. The majority of these reactions have occurred in women, most of whom have a history of allergies.
“The vaccines can safely be given and any adverse reactions that occur acutely have all been relatively easy to manage,” says Dr. Eric Macy, an allergist at Kaiser Permanente in San Diego.

Dr. Kimberly Blumenthal, an allergist at Massachusetts General Hospital, and colleagues published a study that captured the rate of reactions of among almost 65,000 Mass General employees following their first dose of the Pfizer or Moderna vaccine. The researchers identified 16 cases of anaphylaxis, a rate of .025 percent, which included 10 people who reported a history of allergic reactions. Everyone who had anaphylaxis recovered quickly, and none experienced the more severe anaphylactic shock, she notes.
“We estimate there were about 4,000 people in our study who had a history of severe food or drug allergies,” Blumenthal said in an interview. Within this large group of people vaccinated, “thousands with allergies were vaccinated, and nearly all of them were fine.”
Yet, people are hearing anecdotal reports of “reactions” to the COVID-19 vaccines. In the following report, Allergic Living helps readers to understand the differences among: side effects, allergic reactions, as well as non-allergy-based responses to the vaccines and what might trigger them. As well, what to expect with the second dose.
Also a reminder: the CDC recommends people with a history of anaphylaxis due to any cause be monitored for 30 minutes after a vaccine dose, and that all vaccination sites have epinephrine available. It’s also a good idea to bring your epinephrine auto-injectors, and to ask questions to check that those administering the vaccine are ready to treat allergic reactions.
Allergic Reaction vs. Vaccine Side Effect
While anaphylaxis is rare with the COVID-19 vaccines, other types of reactions can occur.
Side effects
Known as “reactogenicity,” side effects are common with many vaccines, and usually come on a few hours to a day after vaccination. With the three U.S.-authorized COVID-19 vaccines, clinical trial data shows that about 60 percent of people report side effects. The effects include arm soreness at the injection site and/or fatigue, along with one or more of headache, muscle or joint pain, fever, chills or nausea. Side effects usually go away within a few days.
Acute allergic reactions
In the Mass General study, 1,365 people – about 2 percent – reported acute allergic reactions, such as itching, rash, hives, swollen lips or face, or wheezing, chest tightness or shortness of breath. These acute responses come on sooner than side effects, usually within a few moments to a few hours after vaccination.
But such symptoms to the mRNA vaccines may not be the type of allergic reaction that’s set off by IgE antibodies. Experts note that there are different types of allergic reactions, and the triggering cause with these vaccines is still unknown. Blumenthal, whose team at Mass General has been at the forefront of coronavirus vaccine allergy research, says there can be triggers that are immunologic, but not allergy-based.
Mast cell responses
A possible explanation for some reactions is that nanoparticles in the mRNA vaccines could set off the immune system’s “complement system”. That’s the part of the immune system normally involved with getting rid of harmful bacteria. The complement system may trigger the release of mast cells, which can mimic an IgE-mediated allergic reaction. Depending on the severity of the symptoms, antihistamines or epinephrine are used to treat mast cell reactions.
Suspected allergy triggers
The mRNA vaccines are not made with any food or animal product proteins that trigger IgE reactions. Interestingly, of the 16 Mass General employees classified as having anaphylaxis, Blumenthal says: “They clinically presented as anaphylaxis. But whether it was anaphylaxis or something else, we cannot really know.”
A few experts have suggested an allergy to polyethelyne glycol (PEG), an inactive ingredient in the vaccine, could explain some reactions. But Macy isn’t convinced, noting that PEG allergy is “vanishingly rare.”
PEG and its chemical cousin, polysorbate, which is contained in the J&J vaccine, are found in everyday product such as cosmetics, toothpaste and laxatives. “Most people with a PEG allergy already know it,” he says. For those reasons, unless PEG allergy is strongly suspected, Macy recommends that people with a history of severe allergies don’t rush to their allergist to get PEG skin testing.
Hives after COVID vaccine
In the Mass General study, about 244 patients reported getting hives after their first mRNA vaccine dose. Hives can be caused by an allergy, but there are many triggers for them. In the condition known as chronic idiopathic urticaria, people get recurring hives without an obvious cause, although there are many possible triggers, including viral infections, hot or cold temperatures, and stress.
By activating the immune system, the COVID-19 vaccine could bring on a bout of hives in people already diagnosed with chronic hives, or in those who are prone to it, Blumenthal says. Chronic urticaria is often managed with antihistamines, although in more difficult cases patients may be prescribed the biologic drug omalizumab (Xolair).
Macy has advised some patients who felt itchy or had a mild case of hives after the first vaccine dose to take an antihistamine, such as cetirizine, before getting the second shot. So far, all of his patients who have done this have received the second dose without an issue. (Speak to your own allergist before doing this though, as there is differing advice).
Vaccine Rash and Emotional Impact
The ‘nocebo effect’
In the placebo effect, people may report feeling better even when they’ve received a sugar pill instead of an actual treatment. Think of the nocebo effect as the placebo effect’s evil twin. When people have negative expectations – such as anticipating side effects or a reaction to a treatment (in this case, the vaccine) – they may experience them.
“Being vaccinated is a heightened emotional situation,” Blumenthal says. “You may feel the joy of getting vaccinated, the fear of an allergic reaction or concerns over adverse effects. That can physically manifest.”
The symptoms aren’t in people’s “heads.” Objective symptoms such as flushing, racing heart and high blood pressure can all be brought on by emotions. “Stress can also cause hives and itching,” says Blumenthal, an expert in drug allergy research who often conducts drug challenges to help her patients determine if childhood penicillin allergies were misdiagnosed (they often are). “I tell my patients, ‘I become itchy in my clinic every week because I talk about it a lot.’”
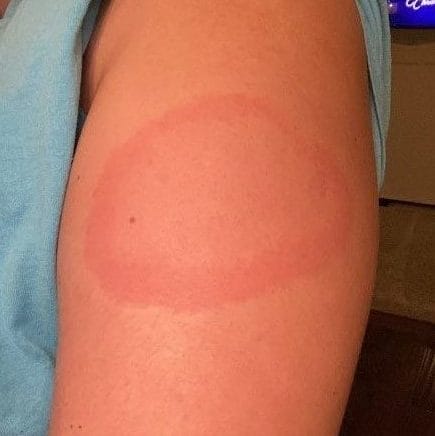
Understanding rash of ‘COVID arm’
Delayed skin reactions, or an angry red rash around the site of the vaccine dubbed “COVID arm,” have also cropped up. In an article for the New England Journal of Medicine, the Mass General researchers described 12 patients who developed a red, tender and sometimes itchy welt on their arm, starting an average of about a week after their first Pfizer or Moderna shot. One also had a rash on the palm and fingers, while another had an elbow rash.
While a rash can be associated with an allergy, delayed skin reactions can simply be a sign that the immune system is kicking into gear. “It can be a normal immune response,” Blumenthal says. In other words, the rash looks bad, but it’s harmless.
Antihistamines and a topical steroid cream got rid of the rash in a few days. One person required a short course of prednisone. All of the patients received their second vaccine dose, and fewer developed the rash the second time.
2nd Shot: Allergic Reaction vs. Side Effect
Second shot experiences
There is no difference between the first and second shots of the Pfizer and Moderna vaccines. But the side effects, such as fatigue, muscle pain, headache and fever and chills, do tend to be more intense after the second.
Yet there is good news too – Macy says that, anecdotally, he is seeing fewer second-dose acute allergic reactions.
More of the ordinary side effects after the second dose are to be expected. The first mRNA shot primes the immune system to recognize and respond to the virus. The second dose acts as a booster to ensure the immune system is ready to mount a full-blown attack.
Though you may feel lousy for a day or two after that second jab, side effects are a sign your immune system is doing its job. Younger adults, who have more vigorous immune systems, report more side effects than older adults. And if you feel fine after your second vaccine, that’s no cause to worry – not everyone experiences side effects.
To treat side effects like muscle pain or fever, the CDC says it’s fine to take acetaminophen or ibuprofen. But don’t take them before the shot, because they might curb the immune system response to the vaccines.
As for fewer second-dose acute reactions, this may provide clues about what’s causing them. If the reactions were IgE-mediated allergies, you’d expect to see more after the second dose, because the body would have been sensitized, says Macy. He has collected data on more than 390,000 Kaiser patients who received the vaccine. “We are seeing very few second-dose reactions thus far,” he notes.
Update: the Kaiser Permanente study is now released here. Among key conclusions in this large population: Only two Moderna vaccinations (0.00033%) resulted in anaphylaxis. Only six of 77 (7.8%) with first-dose reactions to mRNA vaccines also had second-dose reactions.
Related Reading:
FAQ: Allergy Concerns and the mRNA Vaccines
Allergists’ Video: COVID-19 Vaccines and Reaction Risks
The Truth About Drug Allergies





